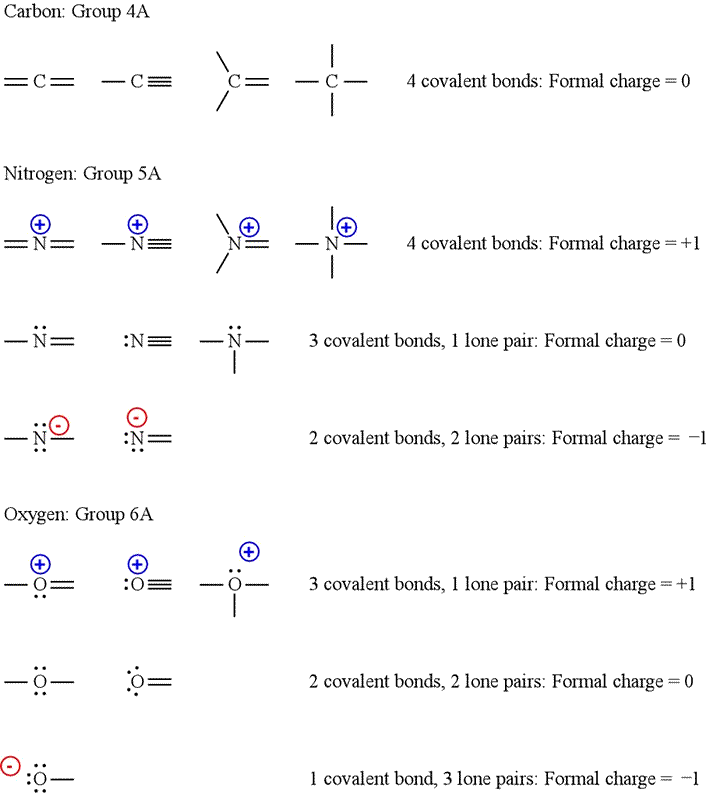
Subtract the number of electrons in the circle from the group number of the element (the Roman numeral from the older. It also gives us a quick estimate of reactivity.Īs an example, second period elements do not exceed the Octet Rule, so you know your structure is invalid if you have 14 electrons on an oxygen atom.īoron compounds often do not have complete octets on boron (for example $\ce$ - 8 electrons, formal charge 2-, oxidation number -2. Alternative Method Count up the number of electrons in the atom's 'circle.' Since the circle cuts the covalent bond 'in half,' each. The Octet Rule (and the related 18 electron rule for some transition metal compounds) serves to give us some idea of whether the structure proposed meets quantum mechanical requirements for valid structures. Formal Charge: Formal charge is the charge that an atom contains. Vocabulary for How to Calculate Formal Charge. CHEMICAL BONDING Calculating formal charge A student proposes the following Lewis structure for the isocyanate (Nco ion. Step 1: Draw the Lewis Dot Structure for the compound given in order to.  
We want to keep track of electrons because it gives us an idea of what type of chemical behavior might be attributable to an atom or molecule. How to Calculate Formal Charge Steps for How to Calculate Formal Charge. in Australia The amount of GST you will pay or should charge customers. As Jan hinted in the comments, there are multiple electron bookkeeping methods which serve different uses. Count the electrons around the atom: Count the number of bonds attached to the atom: Add the number of electrons to the number of bonds attached to the atom and. Calculate the GST (goods and services tax) in Australia with our free GST.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
